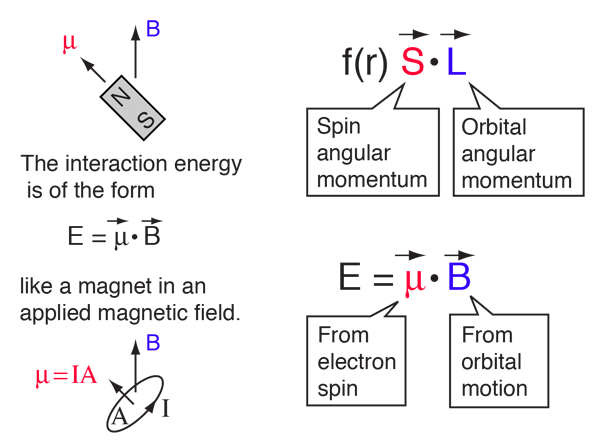
homework and exercises - Calculating the magnetic field of the Proton in Spin-orbit Coupling in Hydrogen Atom - Physics Stack Exchange

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (34 of 78) Magnetic Moment in Hydrogen - YouTube

Three protons coming from excited atomic hydrogen sample are picked up. Their energies are 12.1 eV, 10.2 eV and 1.9 eV . These photons must come from

Magnetic moment due to the motion of the electron in n^(th) energy state of hydrogen atom is proportional to :

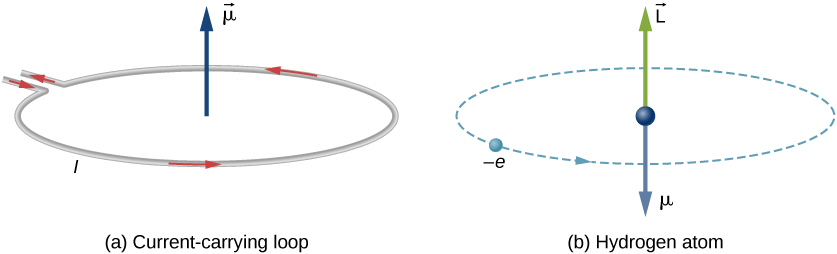
quantum mechanics - Ignoring spin, what is its orbital magnetic moment of an electron in a hydrogen atom in the 2p orbital? - Physics Stack Exchange
Magnetic Moment Physics Of Magnetic Resonance Imaging Magnetic Field Hydrogen Atom, PNG, 1233x874px, Magnetic Moment, Area,

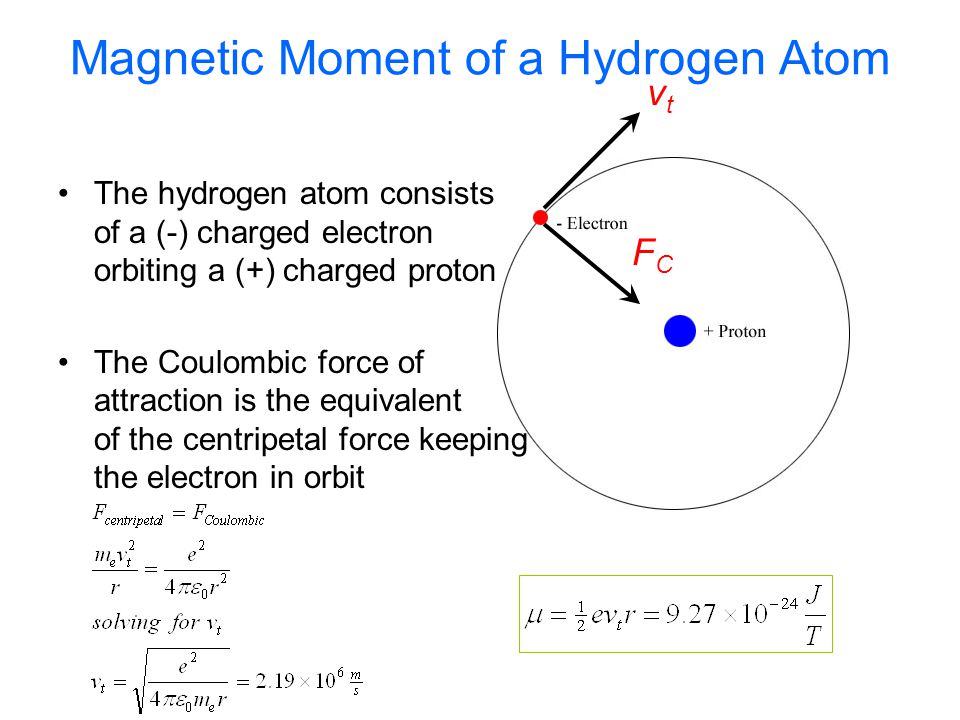
The ratio of magnetic dipole moment of an electron of charge 'e' and mass 'm' in Bohr's orbit in hydrogen atom to its angular momentum is

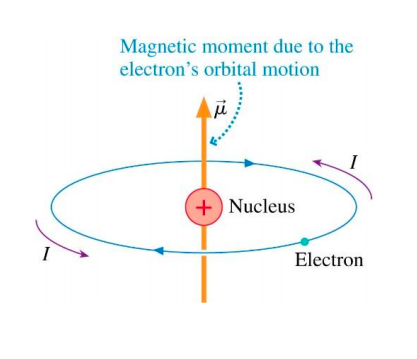
An electron in the ground state of hydrogen atom is revolving in anticlock-wise direction in a circular orbit of radius R. (i) Obtain an experssion for the orbital magnetic dipole moment of

In a hydrogen atom, the binding energy of the electron in the ground state is E1. Then the frequency of revolution of nth electron in the nth orbits is
In a hydrogen atom, an electron of charge revolves in an orbit of radius r with speed v. What is the magnitude of the resulting magnetic moment of the electron? - Quora